Anmol Chemicals is the pioneer manufacturers of Manganese Gluconate, Pharmaceutical Excipients Food & Flavor chemicals in India. We offer Halal and Kosher Manganese Gluconate made in an ISO9001, ISO22000 (FSSC22000) cGMP and GLP certified facility. Our group has several manufacturing facilities spread across the world, supported by toll manufacturers and representatives in UAE, Europe, Africa, USA, China and has several associated manufacturing facilities spread across India. All the Information on Physics, Chemistry, Applications, Uses and Technology on Manufacture of Manganese Gluconate is in these pages. |
| The units have one or more of the certifications like FDA GMP, ISO 9001, ISO 22000, HACCP, REACH, Kosher & Halal |




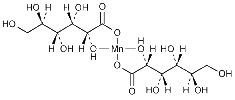
Manganese Gluconate USP Pure & FCC Food Grade Manufacturers
SDS GHS MSDS Sheet of Manganese Gluconate
Manganese Gluconate USP Grade
C12H22MnO14 (anhydrous) --- 445.23
Bis(D-gluconato-O1,O2) manganese.
Manganese D-gluconate (1:2).
Dihydrate --- 481.27
Manganese Gluconate is dried or contains two molecules of water of hydration. It contains not less than 98.0 percent and not more than 102.0 percent of C12H22MnO14, calculated on the anhydrous basis.
Identification—
A: A solution (1 in 20) responds to the tests for Manganese.
B: Chromatographic plate test.
Water: Where labeled as the dried form: between 3.0% and 9.0%, the determination being performed by stirring the mixture containing the Test preparation, maintained at a temperature of 50 , for 30 minutes before titrating with the Reagent. Where labeled as the dihydrate: it is between 6.0% and 9.0%.
Chloride: A 1.0-g portion shows no more chloride than corresponds to 0.70 mL of 0.020 N hydrochloric acid (0.05%).
Sulfate: A 2.0-g portion shows no more sulfate than corresponds to 4.0 mL of 0.020 N sulfuric acid (0.2%).
Limit of lead: To pass the test (0.001%).
Heavy metals: the limit is 20 @g per g.
Reducing substances: the limit is 1.0%.
Manganese Gluconate FCC Food Grade
C12H22MnO14·2H2O Formula wt, dihydrate 481.27
C12H22MnO14 Formula wt, anhydrous 445.24
CAS: [6485-39-8]
DESCRIPTION
Manganese Gluconate occurs as a slightly pink powder. The anhydrous form is obtained by spray-drying, and the dihydrate is obtained by crystallization. It is very soluble in hot water and is very slightly soluble in alcohol.
Function: Nutrient.
REQUIREMENTS
Identification:
A. A 1:20 aqueous solution gives positive tests for Manganese.
B: Chromatographic plate test.
Assay: Not less than 98.0% and not more than 102.0% of C12H22MnO14, calculated on the anhydrous basis.
Arsenic: Not more than 3 mg/kg.
Lead: Not more than 2 mg/kg.
Reducing Substances: Not more than 1.0%.
Water: Anhydrous: Between 3.0% and 9.0%; Dihydrate: Between 6.0% and 9.0%.
For Original Monographs of IP Indian Pharmacopoeia BP British Pharmacopoeia USP US Pharmacopoeia FCC Food Grade product, please check with the respective web-pages or books.
We also offer: Manganese Ascorbate, Manganese Aspartate, Manganese Chloride, Manganese Fumarate, Manganese Lactate, Manganese Lactate Gluconate, Manganese Glycerophosphate, Manganese Sulfate.
Manufacturers
Anmol Chemicals
S-8, SARIFA MANSION, 2ND FLANK ROAD, CHINCHBUNDER, MUMBAI 400009, INDIA
TEL: (OFFICE) 91-22-23770100, 23726950, 23774610, 23723564. FAX: 91-22-23728264
e-mail: ask(at the rate @)anmol.org

Exports to USA, Canada, UAE, Dubai, South Africa, Tanzania, Kenya, Nigeria, Egypt, Uganda, Turkey, Mexico, Brazil, Chile, Argentina, Europe Netherlands, Italy, Spain, Germany, Portugal, France, Malaysia, Indonesia, Thailand, Vietnam, Korea, Japan, etc.
Copyright and Usual Disclaimer is Applicable. 13 September, 2025